Charting a feasible course to a sustainable future
Even as targets are set and pledges made, humanity’s greatest industrial undertaking – the green energy transition – continues to represent an enormous logistic and economic challenge.
We envision a solution rooted in launching the Power-to-X era, enabling electrification of hard-to-abate sectors to support low-carbon chemical and fuel production around the world – at a competitive price point – as well as enabling green steel production and energy supply.
More importantly, we have the technology to make that vision a reality.
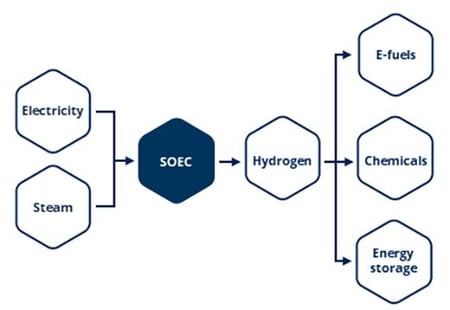
High-temperature electrolysis, made possible by the power of Topsoe’s proprietary solid-oxide electrolysis cell (SOEC), empowers producers in hard-to-abate sectors to generate carbon-free hydrogen or carbon monoxide using renewable electricity. The unmatched efficiency and industrial scalability of SOEC technology position it as an ideal means to an ideal end: satisfying the world’s energy demands with zero negative environmental impact.
Ready to have a conversation about SOEC? Reach out to one of our experts today.
How does SOEC work?
The Topsoe SOEC electrolyzer is a compact stack built primarily from abundant, low-cost ceramic materials enclosed within a metal housing. To produce hydrogen, it utilizes electricity to split water molecules (H2O) into hydrogen (H2) and oxygen (O2). This is accomplished by three components: an anode, a cathode, and an electrolyte. The cathode splits water molecules, via reduction, into hydrogen and oxide ions, after which the oxide ions are transported through the electrolyte to the anode and oxidized into oxygen.
The SOEC unit’s high degree of automation and auto-response capability, across the entire hydrogen process, minimize oversight and training requirements – as well as associated risks – for efficient, convenient on-site hydrogen production and supply.
See how high-temperature electrolysis, made possible by the power of Topsoe’s proprietary solid-oxide electrolysis cell (SOEC), empowers producers in hard-to-abate sectors to generate carbon-free hydrogen or carbon monoxide using renewable electricity.
Transforming the scale of electrolytic hydrogen production
Less than 0.1% of today’s hydrogen is produced through electrolysis, but the increasing availability of renewable energy, at a lower cost, is set to alter the production landscape. By leveraging improved availability with an ultra-high-efficiency Topsoe SOEC electrolyzer, which operates at temperatures exceeding 700°C, up to 30% more hydrogen can be delivered from the same electricity volume at a 30% lower cost

Topsoe SOEC technology’s scalable performance accommodates any plant’s flexibility requirements – whether integrated into industrial processes or used as a standalone unit.
Driving greater electrochemical availability
Topsoe SOEC technology is designed to integrate seamlessly with downstream processes, so green hydrogen can be processed into green ammonia, for use in chemical applications or energy storage; eMethanol, for use in chemical or transportation-fuel production; or one of many other green chemicals and fuels
With decades of experience as a world-leading solution provider for all of these applications, Topsoe is one of the few companies capable of providing the insights and technology needed to facilitate the production and large-scale availability of next-generation fuels and chemicals. We are already engaged in several projects aimed at proving the viability of SOEC, including the NEOM project, announced in July 2020, for which we will deliver the world's largest green ammonia plant.
Unlocking the green potential of CO2
Green hydrogen is a cornerstone of the sustainable future, but the capabilities of Topsoe SOEC technology also extend into the production of green carbon monoxide from captured CO2.
CO2 emissions from waste incineration, power plants, cement production, and industrial facilities can be converted into syngas, a key component in the production of many chemicals and fuels. Syngas is normally produced from natural gas through more carbon-heavy processes, so the use of high-temperature electrolysis helps control or even reduce atmospheric CO2 presence while also ensuring syngas availability for crucial applications. And since the process is entirely electrical, the sole byproduct is pure oxygen.
Enabling the energy transition
A successful energy transition must include solutions for so-called hard-to-abate sectors. Heavy industry and long-haul transportation are responsible for a significant share of global greenhouse gas emissions – and they cannot be electrified directly. So new solutions are needed to decarbonize these vital sectors. Green hydrogen can be used directly as a fuel for transportation or to replace fossil fuels in energy-intensive industries like chemicals, cement, and steel production. It is also a green feedstock for our solutions to produce fuels and chemicals.
